Diagnostics
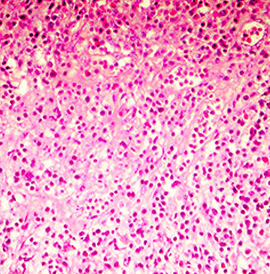
Histopathology
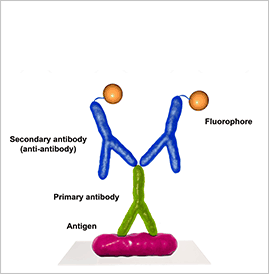
Immunohistochemistry (IHC) test
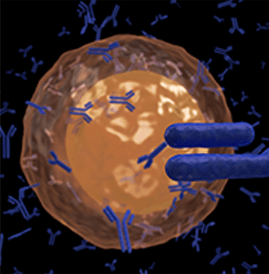
Immunofluorescence (IF)
Immunofluorescence is a microscope-based technique used clinically to diagnose certain cutaneous diseases by detection of autoantibody-antigen complexes.
- Techniques including direct immunofluorescence, indirect immunofluorescence, and salt-split skin are utilized depending on the clinical scenario.
- Direct immunofluorescence is performed on patients’ skin using fluorophore-labeled antibodies that directly bind to the pathogenic autoantibody-antigen complexes in the skin.
- Indirect immunofluorescence techniques are used in dermatology primarily to detect circulating pathogenic autoantibodies.
Fluorescence in situ hybridization (FISH)
It is a molecular cytogenetic technique that uses fluorescent probes that bind to only those parts of the chromosome with a high degree of sequence complementarity. It was developed by biomedical researchers in the early 1980s[1] and is used to detect and localize the presence or absence of specific DNA sequences on chromosomes. Fluorescence microscopy can be used to find out where the fluorescent probe is bound to the chromosomes. FISH is often used for finding specific features in DNA for use in genetic counseling, medicine, and species identification.[2] FISH can also be used to detect and localize specific RNA targets (mRNA, lncRNA, and miRNA) in cells, circulating tumour cells, and tissue samples. In this context, it can help define the spatial-temporal patterns of gene expression within cells and tissues. FISH is used as a confirmatory test for major cancer drugs like Herceptin and other new generation targeted therapies